포함 통증 완화 패치 gaining popularity in the global healthcare market, OEM clients are seeking ways to develop effective, 안전하며, and export-ready products. This guide walks you through the essentials of dosage forms, ingredient selection, and international compliance for a competitive OEM strategy.
✅ 1. Choosing the Right Dosage Form
Each patch type offers unique benefits and fits specific markets:
-
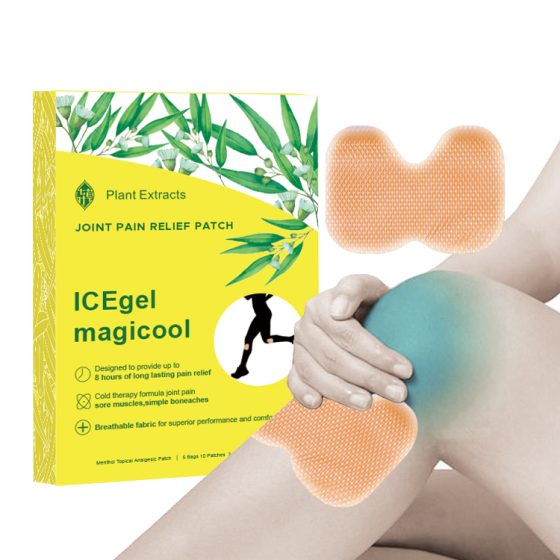
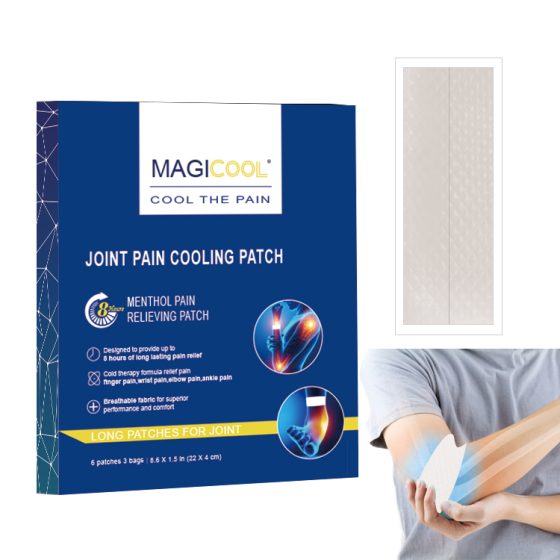
Hydrogel-based: 쿨링, breathable—preferred in Japan and Southeast Asia.
-
Rubber plaster (traditional): High adhesion and warming effect, suited for chronic pain relief.
-
Self-heating patches: Use iron powder to generate warmth and stimulate circulation.
-
Non-woven coated types: Affordable and mass-producible, popular in Europe and North America.
Select based on end-user habits and regional preferences.
🌿 2. Ingredient Strategy for Noticeable Relief
An effective formula should deliver quick results and user comfort:
-
Pain relief actives: 캡사이신, methyl salicylate, menthol.
-
Herbal blends: Mugwort, angelica, dong quai—good for TCM-style products.
-
Soothing agents: Lavender, tea tree—enhance sensory and emotional experience.
For Western markets, label claims like “NSAID-free” and “botanical-based” boost appeal.
🌍 3. Export Compliance & Documentation
-
U.S.: Most pain patches fall under OTC monograph regulations by the FDA.
-
EU: Require MDR compliance and CE certification with strong labeling accuracy.
-
Japan/Korea: Expect rigorous patch tests and INCI-compliant ingredient lists.
OEM factories should have GMP and ISO13485 certifications to meet global expectations.
 Biours 생명 과학 Co., 주식 회사.
Biours 생명 과학 Co., 주식 회사.